
Health is a crown on the heads of the healthy that only the sick can see.
🧬 Fasting and Cancer Research 2026 : What Science Really Shows
🔬 Explore the latest scientific research on fasting and cancer in 2026. Learn how fasting affects tumor metabolism, chemotherapy response, and integrative oncology strategies.
FASTINGCANCERGENERAL
Dr Hassan Al Warraqi
3/14/202615 min read
🧬 Fasting and Cancer Research 2026 : What Science Really Shows
🧬 Fasting and Cancer Research 2026 | Evidence-Based Integrative Oncology Guide
🔬 Explore the latest scientific research on fasting and cancer in 2026. Learn how fasting affects tumor metabolism, chemotherapy response, and integrative oncology strategies.
The Complete Guide to Metabolic Therapy in Oncology
From niche hypothesis to clinical practice — the latest breakthroughs in fasting-based cancer therapy, immunotherapy synergy, fasting-mimicking diets, and precision metabolic oncology.
From Ancient Practice to Precision Medicine
For decades, the idea that "starving" a tumor could treat cancer was dismissed as a dangerous oversimplification.
Critics argued that malnutrition would only weaken the patient, accelerating disease progression rather than stopping it.
However, as we stand in March 2026, the narrative has shifted dramatically.
What was once a fringe hypothesis has evolved into a rigorous field of Precision Metabolic Oncology, backed by robust Phase II and III clinical trials, mechanistic clarity, and a growing consensus among leading cancer centers worldwide.
The turning point wasn't just more data; it was a fundamental change in how we understand the biology of fasting.
We now know that short-term, strategic fasting does not simply deprive the body of energy.
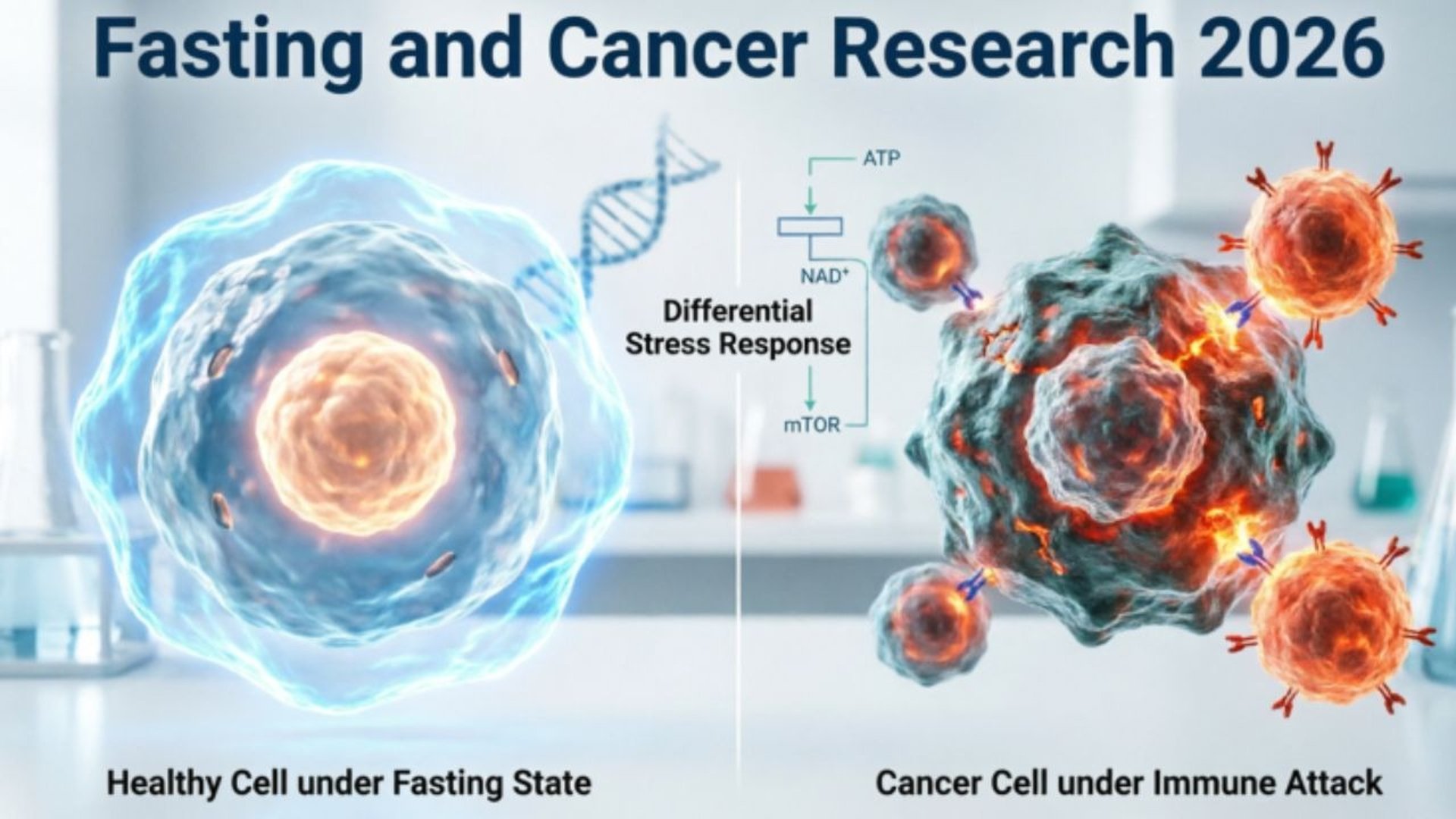
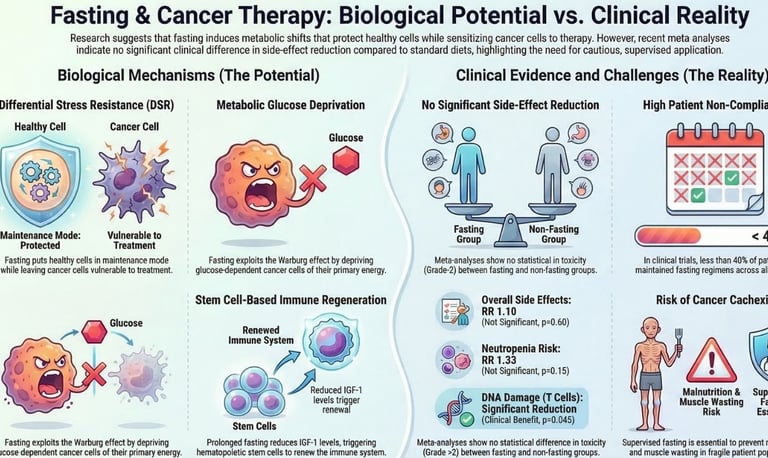
Instead, it triggers a sophisticated biological phenomenon known as the Differential Stress Response (DSR).
In this state, healthy cells enter a protective, dormant mode—shielding themselves from the toxic collateral damage of chemotherapy and radiation—while cancer cells, genetically locked into a state of uncontrolled growth, are left exposed and vulnerable.
The landscape of 2025–2026 has delivered breakthroughs that were theoretical just five years ago:
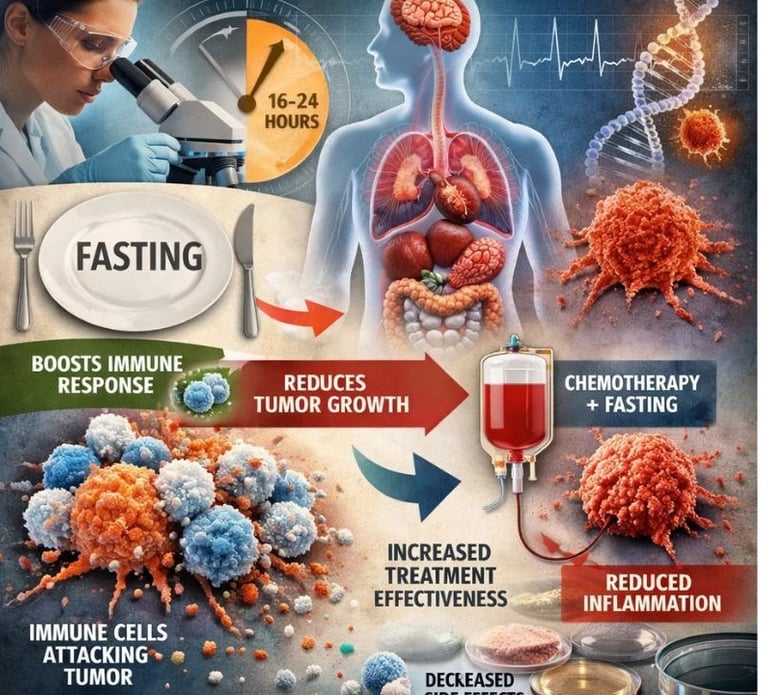
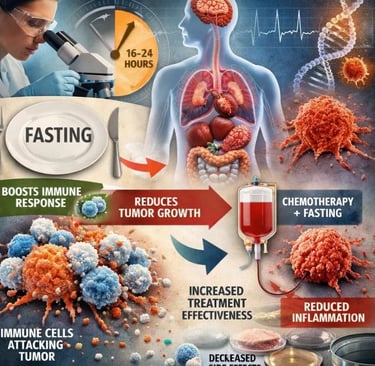
Immunotherapy Synergy: Landmark studies have proven that a mere 16-hour fast can reprogram T-cells, turning "cold" tumors "hot" and significantly boosting the efficacy of checkpoint inhibitors.
Reversing Resistance: New protocols using Fasting-Mimicking Diets (FMD) have shown the ability to overcome drug resistance in ER+ breast cancer by activating specific glucocorticoid pathways.
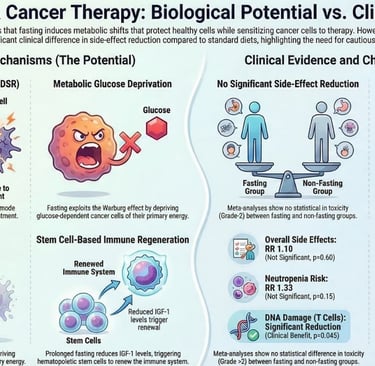
Toxicity Reduction: Large-scale systematic reviews now confirm that fasting protocols can drastically reduce severe side effects like neuropathy, fatigue, and bone marrow suppression, allowing patients to complete full courses of life-saving treatment.
This report cuts through the noise of social media myths and anecdotal claims to present what the science really shows.
We will examine the peer-reviewed evidence from top-tier journals (Cell Metabolism, Nature, The Lancet Oncology), analyze the specific mechanisms at play, review active clinical trials, and outline the critical safety parameters that define modern integrative oncology.
As we move forward, fasting is no longer viewed as an alternative to conventional care, but as a powerful adjunct therapy—a metabolic lever that, when pulled at the right time and under strict medical supervision, can tip the balance in favor of the patient.
Welcome to the new era of metabolic cancer therapy.
Executive Summary: What's New in 2025–2026?
The science of fasting and cancer has undergone a fundamental transformation.
Research through early 2026 has moved fasting protocols from preclinical animal models into robust Phase II and III human clinical trials across multiple cancer types.
The emerging consensus among oncology researchers is clear: strategic fasting is not simply about 'starving' tumors.
It is a precise metabolic intervention capable of reprogramming immune cells, sensitizing tumors to treatment, and shielding healthy tissue from the toxic side effects of chemotherapy.
Below are the most significant breakthroughs from the past 18 months:
Immunotherapy Boost: A landmark study in Cell Metabolism (February 2026) demonstrated that a single 16-hour fast before immunotherapy significantly increases cytotoxic T-cell activity in colorectal cancer patients by altering intracellular levels of the amino acid isoleucine — a finding that may reshape how checkpoint inhibitor protocols are designed.
Survival Rates: Research published in early 2026 showed a 20% improvement in survival in preclinical colorectal cancer models when fasting was integrated with standard-of-care treatment, attributed to slowed tumor proliferation and enhanced drug uptake into tumor cells.
Breast Cancer Breakthrough: New clinical data confirms that fasting-mimicking diets (FMDs) activate glucocorticoid receptors in ER+ breast cancer, effectively delaying or reversing resistance to cornerstone endocrine therapies such as tamoxifen and aromatase inhibitors.
Safety Confirmed: Multiple systematic reviews published in 2025 now confirm that short-term fasting during chemotherapy is safe, clinically feasible, and significantly reduces common adverse effects — including fatigue, nausea, and peripheral neuropathy — without causing clinically significant malnutrition in supervised oncology settings.
Immune Remodeling: Landmark work from Memorial Sloan Kettering Cancer Center showed for the first time that fasting reprograms the metabolism of natural killer (NK) cells, enhancing their ability to survive and function within the hostile tumor microenvironment.
Key Takeaway
Fasting is now considered a legitimate adjunct therapy in integrative oncology — not a fringe idea.
The strongest evidence exists for fasting-mimicking diets combined with chemotherapy or immunotherapy in breast and colorectal cancers.
Patients should always pursue these protocols under direct medical supervision.
The Science: How Fasting Fights Cancer
Fasting does not act through a single mechanism.
Instead, it triggers a coordinated cascade of systemic physiological changes that researchers have termed the Differential Stress Response (DSR).
Under DSR, healthy cells recognize the fasting signal and enter a conserved, stress-resistant protective state — dramatically reducing their vulnerability to the off-target damage inflicted by chemotherapy or radiation.
Cancer cells, by contrast, are often genetically locked into high-growth, nutrient-hungry metabolic states.
Unable to downshift when fuel is scarce, they remain exposed and increasingly vulnerable to treatment.
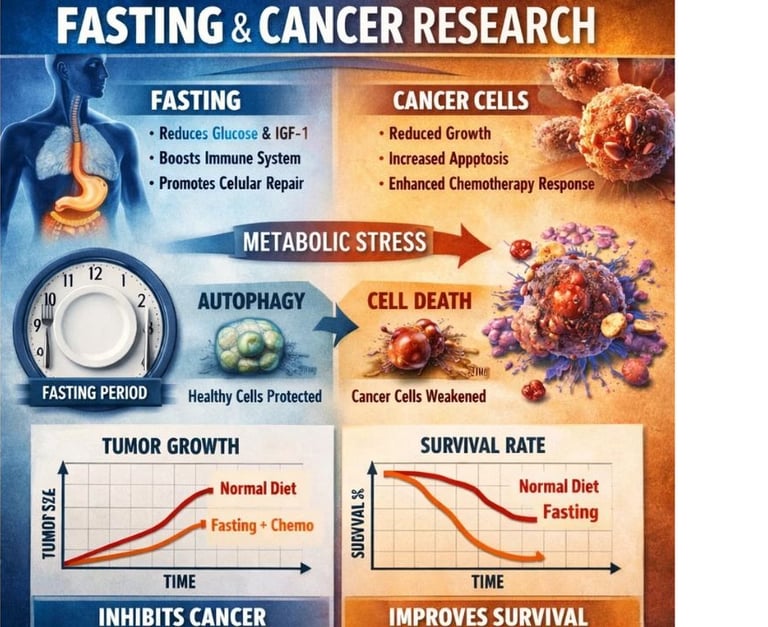
1. Metabolic Reprogramming and the Warburg Effect
Cancer cells are hardwired to consume enormous quantities of glucose and glutamine to sustain rapid proliferation — a phenomenon known as the Warburg effect.
Fasting directly counters this dependency by sharply lowering circulating blood glucose and suppressing Insulin-like Growth Factor 1 (IGF-1), a hormone that acts as a master regulator of cellular growth signaling.
Normal cells adapt efficiently to this metabolic shift by switching their primary fuel source from glucose to ketone bodies, maintaining healthy function even during periods of food restriction.
Cancer cells, lacking the metabolic flexibility of healthy tissue, cannot make this switch effectively.
The result is a state of metabolic stress that is selectively lethal to the tumor.
2026 Mechanistic Update: Emerging research confirms that fasting-induced metabolic shifts also potently downregulate the PI3K/AKT/mTOR signaling pathway, one of the most commonly hyperactivated growth pathways in human cancers, amplifying the anti-tumor effect beyond simple glucose deprivation.
2. Immune System Reprogramming
Among the most transformative discoveries of 2025–2026 is the depth of fasting's impact on the anti-tumor immune response.
This goes far beyond reducing general inflammation, although that benefit remains significant.
T-Cell Activation via Isoleucine: A 16-hour fast triggers a measurable accumulation of the essential amino acid isoleucine within CD8+ cytotoxic T cells. This accumulation initiates a cascade of epigenetic reprogramming and lipid remodeling within the T cell, effectively upgrading these 'killer cells' into a more aggressive, treatment-resistant, and metabolically durable fighting force against tumors.
Natural Killer Cell Enhancement: Research from Memorial Sloan Kettering demonstrated that fasting reprograms NK cell metabolism, enabling these innate immune cells — often the first line of defense against cancer — to survive and function effectively within the nutrient-depleted, immunosuppressive environment surrounding solid tumors.
Reduced Systemic Inflammation: Fasting consistently lowers systemic inflammatory markers including C-Reactive Protein (CRP) and pro-inflammatory cytokines like IL-6 and TNF-alpha. Chronic inflammation is a well-established driver of tumor progression, metastasis, and resistance to therapy. Reducing this inflammatory milieu creates a tumor microenvironment that is fundamentally less hospitable to cancer growth and spread.
3. Autophagy: The Cellular Detox Mechanism
Autophagy — from the Greek for 'self-eating' — is a fundamental biological housekeeping process in which cells digest and recycle their own damaged, dysfunctional, or unnecessary components. Fasting is widely recognized as the most potent natural inducer of autophagy.
Its role in cancer is nuanced and context-dependent.
In Healthy Cells: Fasting-induced autophagy removes cellular toxins, repairs damaged DNA, recycles structural building blocks, and reinforces overall cellular integrity.
This is believed to be a primary mechanism by which fasting protects healthy tissue from chemotherapy-related collateral damage.
In Cancer Cells: While cancer cells can sometimes exploit autophagy as a survival mechanism, the combination of chemotherapy-induced DNA damage with the cellular stress of fasting can push cancer cells beyond their capacity to use autophagy as a rescue pathway.
The result is cell death (apoptosis) rather than survival.
4. Chemotherapy Sensitization
Perhaps the most clinically impactful mechanism of fasting is its ability to function as a powerful chemo-sensitizer.
By inducing a state of metabolic stress in cancer cells prior to treatment, fasting leaves those cells less capable of activating DNA damage repair pathways — the very pathways that allow tumors to survive and recover from cytotoxic agents like cisplatin, doxorubicin, and carboplatin.
This dual action — making cancer cells more vulnerable to chemotherapy while simultaneously activating a cytoprotective state in healthy cells — may allow oncologists to achieve greater therapeutic effect at existing drug doses, or potentially explore higher, more effective doses with a more acceptable side-effect profile.
This remains an active area of clinical investigation.
Fasting Protocols in Modern Oncology
Not all fasting protocols are interchangeable.
The protocol best suited for a given patient depends on cancer type, treatment phase, nutritional status, and individual goals.
As of 2026, four primary, clinically-investigated approaches have emerged:
Protocol
Description
Best Used For
Key 2025–2026 Finding
Short-Term Fasting (STF)
Water-only fast for 24–72 hours around a chemotherapy cycle.
Reducing chemo toxicity; enhancing drug efficacy.
Reduces bone marrow suppression and DNA damage in healthy cells (iScience, Jan 2026).
Fasting-Mimicking Diet (FMD)
Plant-based, low-calorie (~800–1,100 kcal/day), low-protein — typically 5 days per cycle.
Long-term metabolic reprogramming; supporting hormone therapy.
Delays tamoxifen resistance in ER+ breast cancer; improves survival in TNBC models (Nature, Jan 2026).
Time-Restricted Eating (TRE)
All daily calories consumed within an 8–10 hour window (e.g., 16:8 method).
Maintenance therapy; boosting immunotherapy efficacy.
A daily 16-hour fast boosts T-cell response to checkpoint inhibitors (Cell Metabolism, Feb 2026).
Prolonged Fasting (PF)
Medically supervised water-only fasts of 72–120 hours; used sparingly.
Highly aggressive tumors; experimental protocols only.
Emerging data suggests strong mTOR suppression and autophagy induction in solid tumors.
Important Safety Note
All fasting protocols during active cancer treatment must be conducted under the direct supervision of a board-certified oncologist and a registered dietitian with oncology specialization. Self-directed fasting during cancer treatment carries serious risks, including dangerous malnutrition, muscle wasting (cachexia), and potential interference with treatment efficacy.
Cancer-Specific Research Highlights
The evidence base is not uniform across cancer types.
Below is a summary of where fasting research stands for the most studied malignancies as of March 2026:
Cancer Type
Protocol Focus
Key Finding
Trial Status
Breast (ER+)
FMD + endocrine therapy
FMD activates glucocorticoid receptors, reversing tamoxifen resistance.
Phase II/III ongoing
Breast (TNBC)
FMD + carboplatin
Improved overall survival vs. chemo alone in trial data.
Phase II results 2025
Colorectal
16-hour TRE + anti-PD-1
Fasting remodels T-cell metabolism, dramatically improving immunotherapy response.
Cell Metabolism, Feb 2026
Glioblastoma
Ketogenic + TRE (ERGO2 Trial)
Lowered blood glucose and IGF-1 within tumor microenvironment. Survival benefit pending.
Phase II results 2025–26
Ovarian
16:8 TRE + platinum chemo
Aims to reduce neurotoxicity and fatigue; early results promising.
Phase II ongoing 2026
Pancreatic
FMD (preclinical)
FMD may dissolve dense tumor stroma, improving drug delivery to tumor core.
Preclinical / Phase I
Breast Cancer: The Most Studied Arena
Breast cancer, and particularly its ER+ and Triple-Negative (TNBC) subtypes, remains at the leading edge of fasting oncology research.
The reasoning is mechanistic: both subtypes exhibit well-characterized metabolic vulnerabilities that fasting and FMD protocols can selectively exploit.
TNBC and FMD + Carboplatin: Triple-negative breast cancer lacks the three primary receptor targets (estrogen, progesterone, HER2) that define other breast cancer subtypes, making it particularly difficult to treat with targeted therapies.
FMD's ability to create broad metabolic stress in rapidly dividing cells — combined with the DNA-damaging effects of carboplatin — has shown improved overall survival in clinical trial data published in 2025.
ER+ and Endocrine Therapy Resistance: One of the most pressing challenges in ER+ breast cancer management is the development of resistance to tamoxifen and aromatase inhibitors.
FMD has been shown to activate glucocorticoid receptors within ER+ cancer cells — a mechanism that appears to override or reverse common resistance pathways, potentially restoring the effectiveness of endocrine therapies that have stopped working.
Side Effect Management: Across both subtypes, patients undergoing FMD cycles during neoadjuvant chemotherapy consistently report lower rates of severe fatigue, chemotherapy-induced peripheral neuropathy, and gastrointestinal toxicity compared to those on a standard diet.
Colorectal Cancer: Immunotherapy Synergy
The February 2026 Cell Metabolism study has rapidly become one of the most cited papers in fasting oncology.
Researchers demonstrated that a 16-hour fast preceding immunotherapy with anti-PD-1 or anti-PD-L1 checkpoint inhibitors dramatically improved response rates in colorectal cancer patients.
The mechanism — isoleucine-driven metabolic reprogramming of CD8+ T cells — suggests that the timing and duration of fasting relative to immunotherapy administration may be a modifiable variable capable of meaningfully improving outcomes in checkpoint inhibitor non-responders.
Glioblastoma: The ERGO2 Trial
Glioblastoma (GBM) is among the most treatment-resistant cancers known.
The ERGO2 Trial investigated a combined ketogenic diet with time-restricted eating in GBM patients, a protocol specifically designed to target the tumor's reliance on glucose within the blood-brain barrier environment.
Recent results confirmed that the protocol is feasible and successfully achieved measurable reductions in both blood glucose and IGF-1 levels within the tumor microenvironment.
Whether this translates into survival benefit remains under active investigation, with full results anticipated by late 2026.
A critical practical challenge in GBM is preventing cachexia — patients with advanced brain cancer are at extremely high risk of dangerous weight loss, making nutritional monitoring essential.
Pancreatic Cancer: Dissolving the Stroma
Pancreatic ductal adenocarcinoma (PDAC) is notoriously resistant to chemotherapy, in large part because tumors are surrounded by a dense layer of connective tissue called the desmoplastic stroma, which physically blocks drug delivery to the cancer cells.
Early preclinical research suggests that FMD may alter the composition and density of this stroma, potentially creating channels through which chemotherapy drugs can more effectively reach the tumor core.
This is a highly preliminary finding but represents a potentially transformative avenue for one of oncology's most intractable cancers.
Safety, Risks, and Patient Selection
The clinical enthusiasm for fasting in oncology must be tempered by a clear-eyed assessment of who benefits and who is at risk.
The 2026 expert consensus from integrative oncology bodies emphasizes a strongly individualized approach to patient selection.
Who May Benefit from Fasting Protocols?
Patients who are overweight or obese, who are less susceptible to caloric deficits and whose metabolic environment may be particularly amenable to the normalization effects of fasting.
Patients with solid tumors undergoing chemotherapy or immunotherapy who wish to proactively reduce treatment-related side effects in a supervised setting.
Cancer survivors in the maintenance or surveillance phase seeking evidence-based strategies to reduce the risk of recurrence, particularly those with breast cancer.
Patients with ER+ breast cancer who have developed resistance to endocrine therapies and are seeking to restore treatment sensitivity under oncologist guidance.
Absolute and Relative Contraindications
Do NOT Fast if Any of the Following Apply
These are absolute or strong relative contraindications as defined by the 2026 expert consensus.
Fasting in these populations carries serious risks of harm and must not be undertaken outside of a closely monitored clinical trial setting.
Cachexia or Significant Unintentional Weight Loss: Any patient who has lost more than 5% of their body weight unintentionally in the past 6 months should not undergo fasting protocols outside of exceptional clinical circumstances with intensive nutritional support.
Active Eating Disorders: A personal history of anorexia nervosa, bulimia nervosa, binge eating disorder, or other disordered eating patterns represents a significant contraindication due to the risk of relapse and psychological harm.
Type 1 Diabetes: The risk of dangerous hypoglycemia and diabetic ketoacidosis makes standard fasting protocols unsafe in Type 1 diabetic patients.
Modified protocols under endocrinology co-management may be explored in specialized research settings.
Adrenal Insufficiency: Patients with primary or secondary adrenal insufficiency cannot safely mount the hormonal stress response that fasting requires.
This is an absolute contraindication.
Pregnancy and Breastfeeding: Fasting of any significant duration is contraindicated during pregnancy and breastfeeding due to the essential caloric and nutritional demands of fetal development and lactation.
Extreme Frailty or Performance Status: Patients with an ECOG performance status of 3 or 4, or those deemed clinically frail by validated assessment tools, are generally not candidates for fasting protocols.
The Future of Fasting Oncology: What's Next
As the field matures through 2026 and beyond, research is pivoting decisively toward personalization, integration with precision medicine, and regulatory pathways for fasting-based interventions.
The era of one-size-fits-all fasting recommendations is giving way to a more sophisticated, data-driven approach.
Biomarker-Guided Fasting
The next generation of fasting oncology protocols will be guided in real time by individual metabolic biomarkers.
Oncology teams will monitor blood glucose, ketone levels, IGF-1, insulin, and C-reactive protein to determine the precise fasting duration, caloric restriction level, and macronutrient composition needed to achieve the target metabolic state for each specific patient and tumor type.
This moves fasting from a blunt dietary tool to a precision metabolic intervention.
"Nutritechnology" and FDA Pathways
Dr. Valter Longo at the University of Southern California has pioneered the concept of specific dietary patterns as precisely formulated pharmacological agents — a framework he calls 'nutritechnology.' As evidence accumulates, there is growing momentum within the oncology and regulatory communities for formal FDA evaluation of fasting-mimicking diet protocols as approved adjunct therapies alongside standard cancer treatments.
This would represent a paradigm shift in how diet-based interventions are classified, reimbursed, and integrated into clinical care.
Combination Therapy Frontiers
Promising new trials are exploring synergistic combinations that pair fasting protocols with other metabolically active agents:
Fasting + High-Dose Intravenous Vitamin C: The selective pro-oxidant effect of high-dose IV vitamin C in cancer cells may be amplified by the metabolic vulnerability created by fasting, creating a multi-pronged attack on tumor metabolism.
Fasting + Metformin: The anti-diabetic drug metformin inhibits Complex I of the mitochondrial electron transport chain, creating a cellular energy crisis in cancer cells that may synergize powerfully with fasting-induced glucose deprivation.
Fasting + CAR-T Cell Therapy: Early preclinical work suggests that fasting prior to CAR-T cell infusion may optimize the metabolic fitness of the infused cells, potentially improving engraftment, persistence, and anti-tumor efficacy.
Fasting + Radiation Therapy: Building on the differential stress resistance model, researchers are investigating whether fasting windows around radiation treatment can protect surrounding healthy tissue while sensitizing tumors to ionizing radiation.
Frequently Asked Questions FAQS
Can fasting cure cancer on its own?
No. The current body of evidence, including all Phase II and III trials completed or underway as of 2026, supports fasting exclusively as an adjunct therapy.
It is a tool to enhance the effectiveness and tolerability of standard treatments — chemotherapy, radiation, immunotherapy, and targeted therapy — not a replacement for them.
Any claim that fasting alone can cure or eliminate cancer is not supported by peer-reviewed science.
How long do I need to fast to see a therapeutic benefit?
The optimal fasting duration is protocol- and goal-dependent.
For immunotherapy synergy via T-cell reprogramming, a daily 16-hour time-restricted eating window may be sufficient.
For protecting healthy cells from chemotherapy toxicity, a 24–72 hour supervised water fast or a 5-day FMD cycle is typically used in current clinical protocols. Individual variation — body composition, metabolic health, cancer type, and concurrent medications — means that duration should always be determined in consultation with your oncology team.
Is the Fasting-Mimicking Diet safer than water fasting for cancer patients?
For the majority of cancer patients, yes.
The FMD provides essential micronutrients, vitamins, and a small quantity of calories while maintaining the metabolic state associated with fasting — lower blood glucose, elevated ketones, and suppressed IGF-1.
This makes it more clinically tolerable over multi-day periods, substantially reduces the risk of dangerous hypoglycemia, and helps prevent acute muscle wasting.
For active cancer patients, the FMD is generally the preferred protocol over strict water-only fasting, particularly for outpatient use.
Does fasting interfere with the efficacy of chemotherapy?
The available evidence suggests the opposite.
Research consistently shows that fasting makes cancer cells more metabolically vulnerable to cytotoxic chemotherapy drugs while simultaneously activating a cytoprotective state in normal healthy cells.
This differential effect potentially allows standard drug doses to achieve greater tumor cell kill with fewer systemic side effects.
No well-conducted clinical trial to date has demonstrated that fasting reduces chemotherapy efficacy.
However, patients should always disclose dietary changes to their oncology team, as individual circumstances vary.
What should I look for when choosing an oncologist who integrates fasting protocols?
Look for a board-certified medical oncologist with training or strong interest in integrative oncology, ideally affiliated with an NCI-designated cancer center or academic medical center that has an active metabolic oncology research program.
They should work in close collaboration with a registered dietitian (RD) who specializes in oncology nutrition.
Avoid practitioners who present fasting as a cure or who recommend extended fasting protocols without thorough baseline nutritional assessment.
Conclusion
The era of fasting cancer research has unequivocally matured.
Driven by mechanistic clarity, accumulating clinical trial data, and a rapidly growing body of peer-reviewed evidence, fasting and fasting-mimicking protocols are steadily transitioning from integrative fringe to evidence-based adjunct therapy within mainstream oncology.
The most compelling evidence in 2026 supports three primary applications: FMD cycles integrated with chemotherapy to reduce toxicity and enhance efficacy; time-restricted eating to boost the immune response to checkpoint inhibitor immunotherapy; and FMD as a strategy to delay or reverse endocrine therapy resistance in ER+ breast cancer.
What remains constant across all applications is the essential importance of medical supervision, rigorous patient selection, and ongoing nutritional monitoring.
Fasting is a powerful physiological tool.
In the right patient, at the right time, under the right clinical guidance, the evidence increasingly suggests it can meaningfully improve outcomes. Used carelessly, it carries genuine risks.
For Patients and Caregivers
If you are interested in incorporating fasting into a cancer treatment or prevention plan, bring this guide to your next oncology appointment.
Ask specifically about clinical trials in your area testing fasting or FMD protocols for your cancer type.
The clinical trials database at clinicaltrials.gov is a reliable resource for finding enrolling studies.
Medical Disclaimer
This article is intended for informational and educational purposes only and does not constitute medical advice, diagnosis, or a treatment recommendation.
All information presented reflects the state of published peer-reviewed research as of March 2026.
Individual medical circumstances vary significantly.
Always consult with your qualified oncology team — including your medical oncologist and a registered dietitian specializing in oncology — before making any changes to your diet, nutrition, or treatment plan.
==============================================================================================================================================================================================
fasting and cancer research, fasting cancer therapy, fasting and chemotherapy, metabolic therapy cancer, autophagy cancer fasting, integrative oncology fasting, cancer metabolism fasting, fasting mimicking diet cancer, cancer metabolism therapy, fasting immune system cancer
🧬 Fasting and Cancer Research 2026 : What Science Really Shows
https://www.h-k-e-m.com/-fasting-and-cancer-research-2026-what-science-really-shows
fasting cancer research 2026 | fasting-mimicking diet cancer | intermittent fasting chemotherapy | FMD breast cancer | fasting immunotherapy synergy | Valter Longo fasting cancer | metabolic therapy oncology | time-restricted eating cancer survival | short-term fasting chemotherapy | differential stress response cancer | autophagy cancer therapy | IGF-1 cancer fasting | fasting colorectal cancer immunotherapy | TNBC fasting clinical trial | glioblastoma ketogenic diet fasting | fasting natural killer cells | fasting mTOR cancer | integrative oncology 2026
#FastingResearch #CancerResearch #IntegrativeOncology #MetabolicMedicine #CancerMetabolism #OncologyResearch #MedicalResearch #ClinicalResearch #EvidenceBasedMedicine #BiomedicalScience
=========================================================================================================================================================================================

Get in touch
Address
Cairo Al Rehab
Contacts
+20 109 405 2056
hassanalwarraqi@h-k-e-m.com
